TJM2 could be potential treatment for severe coronavirus
$天境生物(IMAB)$ Target Price: $17.93
On Mar 13, I-Mab announced that it would initiate the development of TJM2 to treat cytokine storm (CS) in patients with severe coronavirus infection.
TJM2, indicated for RA and side effects of CAR-T related therapies, is in Phase I clinical trial in US and could initiate Phase I trial in China in early 2020.
TJM2 could potentially treat severely affected patients of coronavirus through neutralizing increased cytokines.
Initiating the development of TJM2 for the treatment of CS associated with severe coronavirus infection.On March 13, I-Mab announced it has started the development of TJM2 for the treatment of CS in patients seriously sick with coronavirus. According to the company, the study will kick off in the US and expand into other hard-hit countries. The development is now pending investigational new drug (IND) application acceptance by US FDA.
TJM2 now in Phase I trial in US for indications for autoimmune disease and CRS.I-Mab’s TJM2 is one of the company’s Global Portfolio pipeline assets. It is an anti-human-granulocyte-macrophage colony-stimulating factor (GM-CSF) monoclonal antibody (mAb) indicated for: 1) autoimmune disease; 2) cytokine release syndromes (CRS) caused by rheumatoid arthritis (RA); and 3) side effects of CAR-T related therapies. It is now in Phase I clinical trial in the US and I-Mab targets to initiate Phase I trial in China in early 2020.
Targeting CS, TJM2 could be a potential treatment for severe coronavirus patients.CS refers to the phenomenon that a variety of cytokines in body fluids are produced rapidly and massively after organism infection, which is an important cause of acute respiratory distress syndrome and multiple organ failure. A recent research paper published by The Lancet on Jan 24, 2020 ("Clinical features of patients infected with coronavirus in Wuhan, China") stated that coronavirus-affected patients showed a significant increase in proinflammatory cytokines; and on Feb 13, 2020, the clinical team of phama company Dirui Industrial (300396 CH, NC) found that for severely affected coronavirus patients, it is necessary to prevent the occurrence of acute symptoms caused by CS. These research results have provided the basis for a novel concept for treatment of severely affected patients with coronavirus: neutralizing the increased level of cytokines by monoclonal antibodies may prevent deterioration and death, according to Journal of Immunobiology and Immunotherapy on Feb 14, 2020. We think the expanded clinical application of TJM2 by I-Mab may be one factor to help combat the coronavirus pandemic.
Maintain BUY and DCF-based TP of US$17.93. Key risks:clinical trial and commercialization delay; and inability to obtain financing.
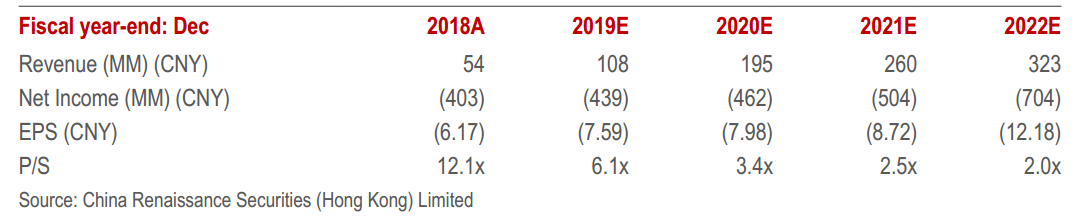
Summary Financial Data
Disclaimer: Investing carries risk. This is not financial advice. The above content should not be regarded as an offer, recommendation, or solicitation on acquiring or disposing of any financial products, any associated discussions, comments, or posts by author or other users should not be considered as such either. It is solely for general information purpose only, which does not consider your own investment objectives, financial situations or needs. TTM assumes no responsibility or warranty for the accuracy and completeness of the information, investors should do their own research and may seek professional advice before investing.
- 菜鸟学炒股ing·2020-03-27英语不太好,这个意思是说,天境生物可以治新冠吗?[财迷]LikeReport
